The psychological toll of chronic and terminal illness
When a patient receives a chronic or terminal illness diagnosis, their lives are likely to change radically. The shock and uncertainty of such a diagnosis often manifest through high levels of anxiety and depression. One study among patients diagnosed with cancer reported that 50% presented symptoms of anxiety.[1] Similar prevalence figures have been reported for chronic respiratory conditions (50%) and chronic pain (46%).[2,3] This reflects a dramatic increase compared with anxiety prevalence among the general population, which is 11.3% in the United States.[4]
Beyond the difficulty of living with a mental health issue, this can also have serious implications for the efficacy of a patient's treatment. For example, patients suffering from anxiety have significantly increased symptom severity scores compared with their non-anxious counterparts and depressed patients are three times more likely to be non-compliant with their treatment regimen.[5,6] The psychological aspects of chronic and terminal diagnoses remain undertreated by modern medicine overall, creating a major gap between the treatment of disease and the management of the patient’s mental health journey.
Insights from placebo effects
The placebo effect is a phenomenon that reflects a situation in which a patient’s symptoms improve despite using a non-active treatment approach. While placebo effects are often received with dismay by researchers conducting clinical trials, they can reveal invaluable insights about the psychological factors that impact a patient’s recovery. One such factor that has been extensively studied is the patient–practitioner relationship.
A study led by Harvard University researchers investigated the therapeutic effects of a mock acupuncture procedure (placebo) on irritable bowel syndrome patients7. Divided into two groups, the patients were given placebo treatment. In one group, the practitioner conducted the treatment in a warm, attentive, and confident manner; in the other group, the practitioner exercised a neutral manner with limited patient engagement. The patients that were treated in the former manner reported a 1.6-fold improvement in symptom severity, compared with a 0.14-fold improvement for when the practitioner adopted a neutral manner.[7]
The above is only one of several examples. The effects of the patient–practitioner relationships on symptom severity have also been reported in studies on gastroesophageal reflux disease, knee osteoarthritis, vasovagal syncope, and multiple sclerosis.[8,9,10,11] By simply exhibiting a supportive, empathetic, and attentive manner, practitioners can improve certain symptoms before administering any active treatment. Questions exist, however, as to how this approach can be implemented in a clinical context; particularly, in an overstretched and chaotic healthcare environment, how can healthcare practitioners feasibly deliver the emotional and psychological support their patients need?
Mobile healthcare for continuous support
Mobile devices have revolutionized almost all aspects of daily life including work, travel, and socializing. The latest estimates indicate that almost 80% of individuals in the US own a smartphone.[12] Furthermore, adults spend a median of 89 minutes per day on their smartphone, equating to 3.7 minutes per hour.[13] Mobile devices are now a central, and potentially dominant component of how people connect with one another, build relationships, and exchange information. These devices also represent an opportunity for providing attentive, continuous patient support outside of the healthcare environment. This is the concept of “mobile health” or “mHealth”, which is a broad term that encompasses the use of mobile devices to manage and administer healthcare. This approach allows healthcare providers to deliver information, educational content, and on-demand support from any location, without the need for direct administration by a physician.
In the first quarter of 2021, there were 53,979 mHealth apps available in the Apple App store, up from just above 28,000 in the first quarter of 2015.[12] These mHealth applications promise major benefits for both the patient and the healthcare provider, with the former having 24/7 access to healthcare support from any location and the tools to confidently manage their illness, keep track of their medication schedule, and receive remote counseling from peers and professionals. Healthcare providers gain a more complete visibility of their patients' symptoms in real-time, population-scale data, and automated patient aftercare. In the sections that follow, we explore some of these mHealth applications in more detail and discuss their efficacy data for improving patient outcomes.
The reSET application for addiction: The first Food and Drug Administration-authorized prescription digital therapeutic
In late 2017, the United States Food and Drug Administration (FDA) approved the reSET application as the first-ever prescription digital therapeutic (PDT). The application was created by Pear Therapeutics (2018), a company specializing in the development of PDTs. Their lead technology is the reSET program for substance abuse, which is marketed in partnership with Sandoz (c/o Novartis).
The application provides a 90-day PDT program that administers cognitive behavioral therapy (CBT) and contingency management to the patient to help maintain abstinence from their addicted substance. For the healthcare provider, the reSET dashboard (see Figure 1) provides access to data about the patient's symptoms and their use of the application including lessons completed, patient-reported substance abuse, patient-reported cravings and triggers, compliance rewards, and urine screen results. The goal of the program is to increase patient abstinence from substances of abuse during their treatment, and to increase the rates of patients completing their treatment program. Pear Therapeutics have reported a more than two-fold increase in abstinence among patients who used the application for addiction to alcohol, cocaine, cannabis, or stimulants, and "significant increases" in treatment retention (n = 400).[14] The company released a second version of the reSET application for opioid use disorder, reSET-O, in late 2018.[15]
Oncology Together, LivingWith, and BeLive: Pharmaceutical aftercare
Pfizer launched two programs to support cancer patients who received treatment using Pfizer Oncology medications and one for diabetes and fibromyalgia patients experiencing chronic pain. The first, Pfizer Oncology Together, provides dedicated social workers, or “care champions”, to assist patients with information about their treatment, provide emotional support resources, assist in workplace transition, and provide help with transportation and financial assistance. The goal of Oncology Together is primarily to aid patients with the practical challenges of initiating cancer treatment and to help ease some of the uncertainty and anxiety surrounding the initial phases of treatment.
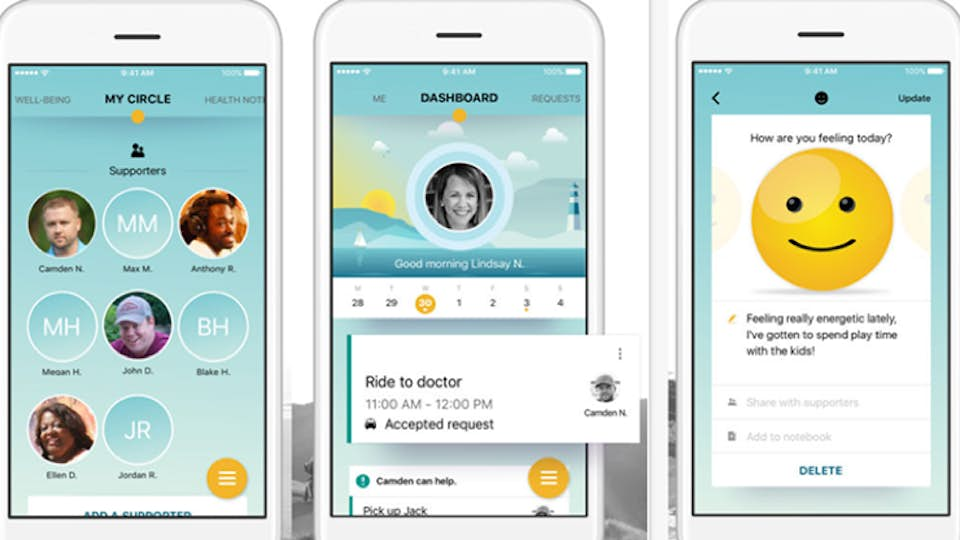
The second app, LivingWith, places a larger focus on day-to-day patient support, with capabilities for establishing peer and family/friend networks, symptom tracking, and information about the individual user’s disease and treatment (see Figure 2). The application also facilitates requests for help from family and friends, such as transportation to a doctor or receiving items from a supermarket. The goal of LivingWith is to help patients manage the daily challenges of living with cancer and to provide a specialized social network focused on supporting the patient in their treatment journey.
The BeLive application is paired with a wristband that is used to log pain levels on a scale of 0 to 10 at intervals throughout the day. Patients also report their anxiety levels and sleep quality, allowing them to share data with their clinicians that can help to better inform their treatment. Early results have shown that 78% of patients who used the application were more likely to comply with their treatment plan. Without developing new medications that infer significant research and development cost implications, the pharmaceutical company was able to improve the treatment outcomes of their patients simply by providing ongoing support.
The PTSD Coach application: remote behavioral therapy
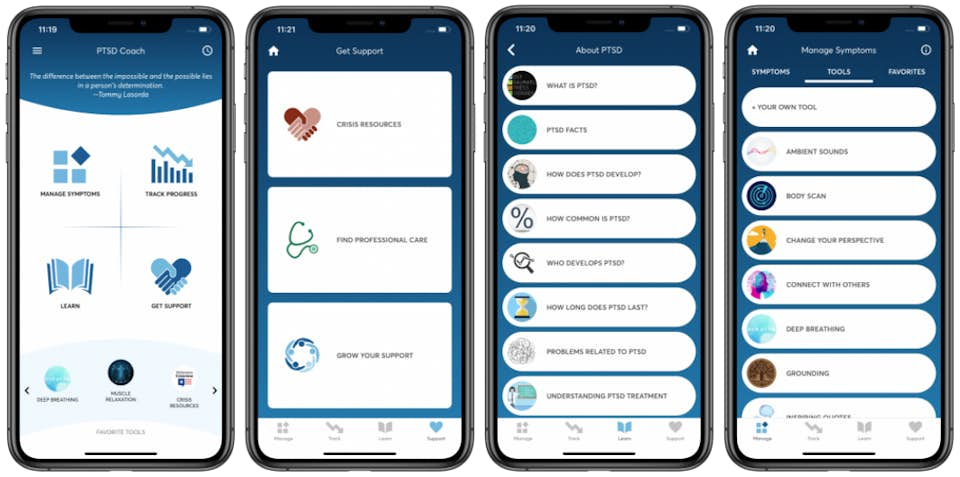
The post-traumatic stress disorder application, PTSD Coach, was launched in 2011 by the U.S. Department of Veteran Affairs. It provides education about PTSD, PTSD self-assessment, resources for finding support, and tools to help users manage their daily life. The primary goal of the application is to improve mental health literacy and potentially increase treatment-seeking behavior among PTSD sufferers who may benefit from evidence-based behavioral therapies, such as CBT. The application includes four key modules, “learn”, “track symptoms”, “manage symptoms” and “get support” (Figure 3). In a pilot study, modest improvements from baseline values were observed in PTSD symptoms, depression, and social quality of life scores among those who used the application.[16]
Country-specific versions of PTSD Coach have been released in Australia, Canada, Germany, and Denmark, and a new version called Cancer Distress Coach is in development for PTSD related to cancer diagnosis and treatment.
Conclusions
The importance of a patient’s mental health when coping with a chronic illness is clear. Being diagnosed with a disease is an anxious and confusing time, and without the right communication and support, patients can easily fall victim to pathological anxiety and depression, which can have devastating effects on their recovery. With high inpatient workloads, healthcare staff is often simply unable to provide adequate outpatient support. In this context, mHealth can make positive contributions. Mobile healthcare provides an efficient means for integrating patient education, self-management training, continuous support, and data-driven healthcare management. To date, the assembled data presents promising results for improving adherence to treatment regimens and even increasing the effectiveness of prescribed drugs. If mHealth applications are found to be efficient and cost-effective at scale, they may become a routine component of medical treatment, to be prescribed by doctors in the same way as an antibiotic for a bacterial infection.
References
1. Linden W, Vodermaier A, MacKenzie R, Greig D. Anxiety and depression after cancer diagnosis: Prevalence rates by cancer type, gender, and age. Journal of Affective Disorders. Vol 141. Elsevier; 2012:343-351. doi:10.1016/j.jad.2012.03.025
2. Bordoni B, Marelli F, Morabito B, Sacconi B. Depression, anxiety and chronic pain in patients with chronic obstructive pulmonary disease: The influence of breath. Monaldi Arch Chest Dis. 2017;87(1):106–112. doi:10.4081/monaldi.2017.811
3. Bair MJ, Wu J, Damush TM, Sutherland JM, Kroenke K. Association of depression and anxiety alone and in combination with chronic musculoskeletal pain in primary care patients. Psychosom Med. 2008;70(8):890–897. doi:10.1097/PSY.0b013e318185c510
4. Strine TW, Mokdad AH, Balluz LS, et al. Depression and anxiety in the United States: Findings from the 2006 Behavioral Risk Factor Surveillance system. Psychiatr Serv. 2008;59(12):1383–1390. doi:10.1176/ps.2008.59.12.1383
5. Katon W, Lin EHB, Kroenke K. The association of depression and anxiety with medical symptom burden in patients with chronic medical illness. Gen Hosp Psychiatry. 2007;29(2):147–155. doi:10.1016/j.genhosppsych.2006.11.005
6. DiMatteo MR, Lepper HS, Croghan TW. Depression is a risk factor for noncompliance with medical treatment meta-analysis of the effects of anxiety and depression on patient adherence. Arch Intern Med. 2000;160(14):2101–2107. doi:10.1001/archinte.160.14.2101
7. Kaptchuk TJ, Kelley JM, Conboy LA, et al. Components of placebo effect: Randomised controlled trial in patients with irritable bowel syndrome. BMJ. 2008;336(7651):999–1003. doi:10.1136/bmj.39524.439618.25
8. Dossett et al. Patient-provider interactions affect symptoms in gastroesophageal reflux disease: A pilot randomized, double-blind, placebo-controlled trial. PLoS One. 2015;10(9). doi:10.1371/journal.pone.0136855
9. Suarez-Almazor et al. A randomized controlled trial of acupuncture for osteoarthritis of the knee: Effects of patient-provider communication. Arthritis Care Res. 2010;62(9):1229–1236. doi:10.1002/acr.20225
10. Pournazari P, Sahota I, Sheldon R. High Remission Rates in Vasovagal Syncope: Systematic Review and Meta-Analysis of Observational and Randomized Studies. JACC Clin Electrophysiol. 2017;3(4):384–392. doi:10.1016/j.jacep.2016.10.012
11. Ehde DM, Elzea JL, Verrall AM, Gibbons LE, Smith AE, Amtmann D. Efficacy of a Telephone-Delivered Self-Management Intervention for Persons With Multiple Sclerosis: A Randomized Controlled Trial With a One-Year Follow-Up. Arch Phys Med Rehabil. 2015;96(11):1945–1958.e2. doi:10.1016/j.apmr.2015.07.015
12. Statista. Healthcare apps available Apple App Store 2021. Published 2021. Accessed June 3, 2021. https://www.statista.com/stati...
13. Christensen et al. Direct Measurements of Smartphone Screen-Time: Relationships with Demographics and Sleep. PLoS One. Published online 2016. doi:10.1371/journal.pone.0165331
14. Pear Therapeutics. Pear Therapeutics Presents New Data on reSET® and reSET-OTM at American Academy of Addiction Psychiatry Annual Meeting and Scientific Symposium - Pear Therapeutics. Published 2018. Accessed June 3, 2021. https://peartherapeutics.com/p...
15. MobiHealthNews. Pear’s digital therapeutic reSET-O FDA cleared to treat opioid use disorder. Published 2018. Accessed June 3, 2021. https://www.mobihealthnews.com...
16. Possemato et al. Using PTSD Coach in primary care with and without clinician support: a pilot randomized controlled trial. Gen Hosp Psychiatry. 2015;38:94–98. doi:10.1016/j.genhosppsych.2015.09.005

Dr Rachel Murkett
Project Director
Dr Rachel Murkett leads strategic intelligence projects at Biochromex. She holds a PhD in Chemistry from the University of Cambridge and has 6+ years of experience as a scientific consultant to consumer goods brands.